Asthma Medication linked to Childhood Suicide
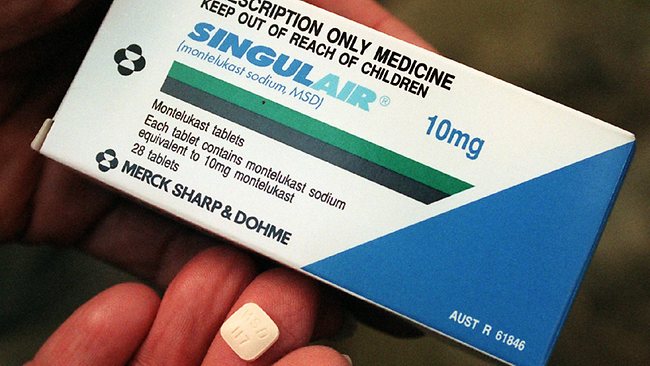 The Therapeutic Goods Administration (TGA) has recently published new public health warnings on a popular asthma medication, Singulair, which is currently used by over 20,000 Australians.
The Therapeutic Goods Administration (TGA) has recently published new public health warnings on a popular asthma medication, Singulair, which is currently used by over 20,000 Australians.
This medication is the cause of 58 separate reports of adverse psychiatric effects in children and teenagers, as serious as suicidal ideation and depression over the last 13 years.
Other reported effects of this medication include altered moods, nightmares, insomnia and agitation.
In fact, many patients taking the medication experienced multiple psychiatric symptoms concurrently.
Although this number in incidents seems fairly low, the TGA explains that “the inherent difficulty in establishing psychiatric diagnoses in children could contribute to under-reporting of related effects to the TGA”.
Treatment outcomes like this aren’t particularly surprising for a medication with product warnings including possible side effects of agitation, aggressive behavior or hostility, anxiousness, depression, disorientation, disturbance in attention, dream abnormalities, hallucinations, insomnia, irritability, memory impairment, restlessness, somnambulism and suicidal thinking.
What is surprising is the fact that Singulair was manufacturing company, Merck’s biggest selling drug worldwide and made around $5 billion profit per year until 2012 when its patent expired, despite it’s potential side effects.
There is now more competition on the market as cheaper generic brands have made the same formula available.
How Does It Work?
Singulair acts as a leukotriene receptor antagonist which blocks specific inflammatory mediators to reduce airway constriction (bronchoconstriction).
Singulair is indicated for asthma prophylaxisis and chronic treatment rather than acute treatment. It is prescribed to adults and children as young as 12 months old, and is often used as an add on to inhaled corticosteroid treatments for asthma.
Other uses for the drug include treatment for allergic rhinitis, seasonal allergic rhinitis and exercise-induced asthma.
Where To Go From Here
As with any medication, it is essential for practitioners to weigh up the risks vs. benefits of the drug and base their prescription on the most safe and efficacious treatments available.
It is also the personal responsibility of anyone choosing to take a medication, or allowing their children to take a medication educate themselves about potential risks of the drug, and to closely monitor any adverse effects.
While it is clear that not everyone experiences psychiatric effects from taking Singulair, studies have found that people with asthma and atopic conditions are more vulnerable to neuropsychotic symptoms than the general population to begin with.
Other factors increasing risk of psychiatric side effects from Singulair may include preexisting mental conditions, a family history of mental illness, additional psychological triggers or susceptibilities to mood disorders.
Once you know the facts, it’s a gamble you may not want to take with your child’s health or your own.
Fortunately, in the case of asthma, there are often many lifestyle, environmental and dietary factors that can be adjusted to safely manage the symptoms of the condition without the use of serious medications.
Indeed there can often be underlying neuro-musculoskeletal complaints that can have a negative impact on your ability to breath and drive asthmatic presentations.
Worth a chat on your next consultation; in practice or in the eClinic.
According to News.com.au:
AN ASTHMA treatment used by 20,000 Australians has been linked to suicidal thoughts in teenagers and children and doctors are being urged to prescribe it with caution.
The nation’s medicines watchdog has revealed it received 58 reports of adverse psychiatric events in children and teenagers being treated with Singulair between January 2000 and January 2013.
These included five reports of suicidal ideation, five reports of depression and eight reports of agitation, the Therapeutic Goods Administration says.
This could be an underestimate of the problem because the TGA says it is difficult to establish psychiatric diagnoses in children.
The watchdog’s warning is included in its latest monthly medicines safety update which also reports other adverse reactions which link nightmares, altered mood and insomnia to the medicine.
”In many cases, patients experienced multiple neuropsychiatric reactions,” the TGA says.
”Health professionals should be aware of these potential adverse effects and advise patients and parents to seek medical advice should they occur.
Asthma Australia medical expert Professor Adam Jaffe says Singulair is used more commonly in treating children than adults and is ”highly effective” in the majority of cases.
”In a very small minority we do see behavioural side effects like nightmares and hyperactivity and in those children we have to stop it,” he said.
”With any medication you have got to balance the risks versus the benefits and Singulair is no different,” he said.
Professor Jaffe said it was his practice to talk to parents about the possible side effects because they usually went straight to the internet to research medicines prescribed for their children.
The drug company Merck includes warnings in its product information of possible side effects including suicidal ideation, depression, agitation, aggressive behaviour, hallucinations, insomnia, somnambulism and tremor,
Singulair was Merck’s biggest selling drug earning around $5 billion a year worldwide until last year when its patent expired in the United States.
Cheaper generic versions of the drug are now available but not yet on the pharmaceutical benefits scheme in Australia.
The TGA wants doctors to carefully evaluate the risks and benefits of continuing treatment with Singulair if psychiatric events occur in a patient.
”In the case of children, caregivers should also be made aware of these potential adverse effects and instructed to seek medical advice if they have any concerns,” the TGA says.
The US Food and Drug Administration investigated Singulair in 2009 and investigated the manufacturer’s clinical trial reports on the drug and found psychiatric problems were not frequently reported but noted the trials were not designed to look for the issue.
The Spinal Centre – Neuro-Musculoskeletal SolutionsThe Spinal Centre provides the Best Natural Medicines, Health Products and Nutritional Solutions to Free You of Pain and Improve Your Spinal Health.The Spinal Centre is a leader in the management and treatment of spinal pain and disability including:
- Chronic Back and Neck Pain
- Cervical and Lumbar Disc Injuries with or without neruological changes
- All forms of arthritis including rheumatoid arthritis, psoriatic arthritis and osteoarthritis
- Neurodegenerative diseases such as Parkinson’s and Alzheimer’s disease
- Neuromuscular problems such as chronic back pain, headaches, migraines, neck or spinal pains
- Myofascial pain syndromes such as fibromyalgia, diffuse muscular or joint pains
- Austistic spectrum disorders including ADD, ADHD, learning difficulties and autism
- Fatigue, energy and mood disorders including chronic fatigue and depression
The Spinal Centre focuses on resolution with the development of individualised treatment programs using Conservative and Integrative Medicine; combining genetic, metabolic and nutritional testing with specific natural medicines to enhance your health and achieve your genetic potential.We have helped thousands – Why Not You?